We use cookies to ensure that we give you the best experience on our website. It will mean you have agreed with we would get cookies when you continue to see this website.

intellim establishes a development support system for practicalapplication of regenerative medicine, including regulatory affairs'consultation, clinical trials, manufacturing, and coordinationw ith KOLs.


Regenerative medicine experts will answer questions about manufacturing, non-clinical and clinicalchallenges, provide proposal-based consultation to solve problems.
Our group is actively participating in the formation of the Osaka International Center for Future Medical Care and is promoting the construction of a regenerative medicine platform standards.
The difference of regulation may be difficult issue in regenerative medicine development. intellim is able to provide constant support for accurate and updatedregulatory compliances including irregular cases.
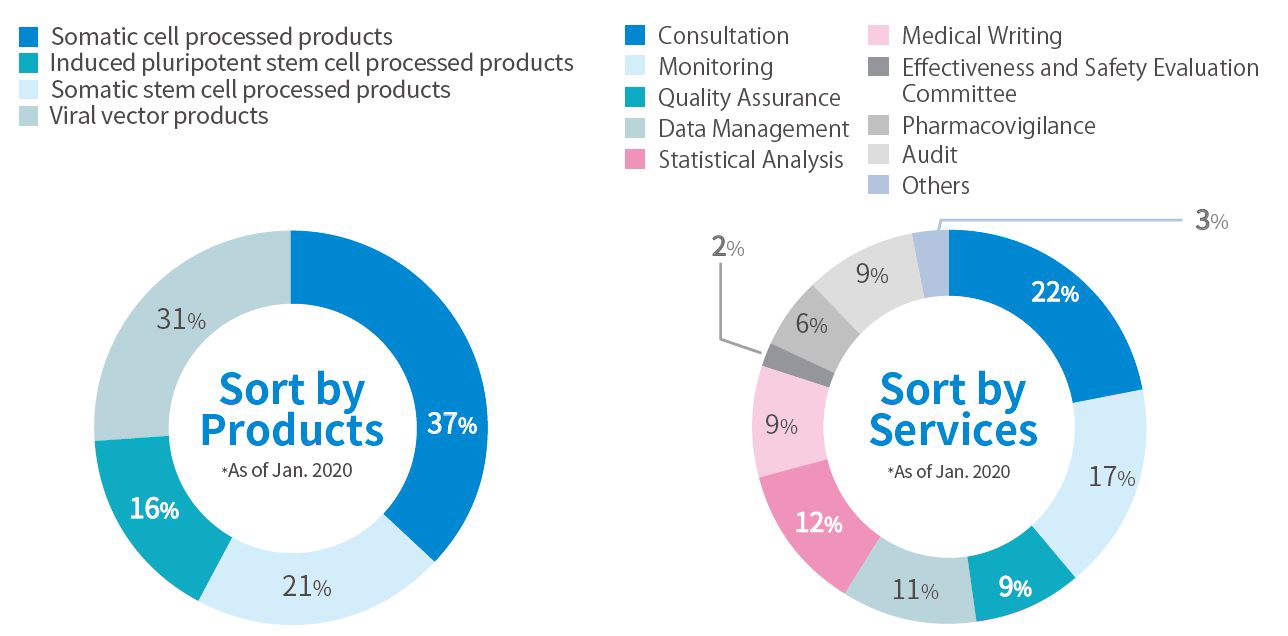
As Experts in the field of Regenerative Medicine, We will propose the most optimal pathway to an Exit Strategy.
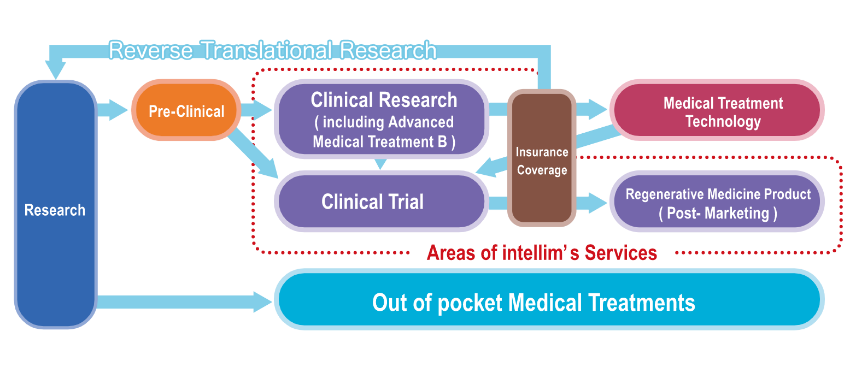
For more information
If you have any comments, questions, please contact us.