We use cookies to ensure that we give you the best experience on our website. It will mean you have agreed with we would get cookies when you continue to see this website.
Our global network will lead your Asian study to success.
intellim is looking toward to be “One-Stop Service Provider” to support clinical development in a
central focus on Japan. intellim supports many pharmaceutical companies who are considering to
conduct Asian studies, and also, we are putting a lot of effort into supporting Bio Venture companies.




Extensive and experienced in trials in Japan and Asia supporting global partners and clienets.
We provide medical writing, regulatory consultation and other essential services for domestic and international
development of pharmaceuticals, medical devices and regenerative medicine products.
intellim can also support “First in Human” outside of Japan.
Especially strong in the country mixed global trials for East and Sounth East Asia.
Experienced both trials from EU and US based clients.
Our contact personnel are multilingual and both fluent in English and Japanese to ensure the communications for
global requests.
They are familiar with regulatory and circumstances of drug development in Asia and other countries will support
you the best proposal for you.
intellim has bilingual (etc. Japanese / English / Korean / Chinese) staff members to help you.
See Our PM's summary of career.
As Japanese CRO, intellim can understand Japanese clients' thought. For this reason, intellim can provide One-Stop Service with high quality and speed for you, which cannot be provided by major Global CRO.
Dr. Nagahiro Saijo who is our special advisor (also special advisor of Japanese Society of Medical Oncology) will support your drug development with his broad KOL Network. He has very good communication with KOLs not only in Asia, but also in US/Europe.
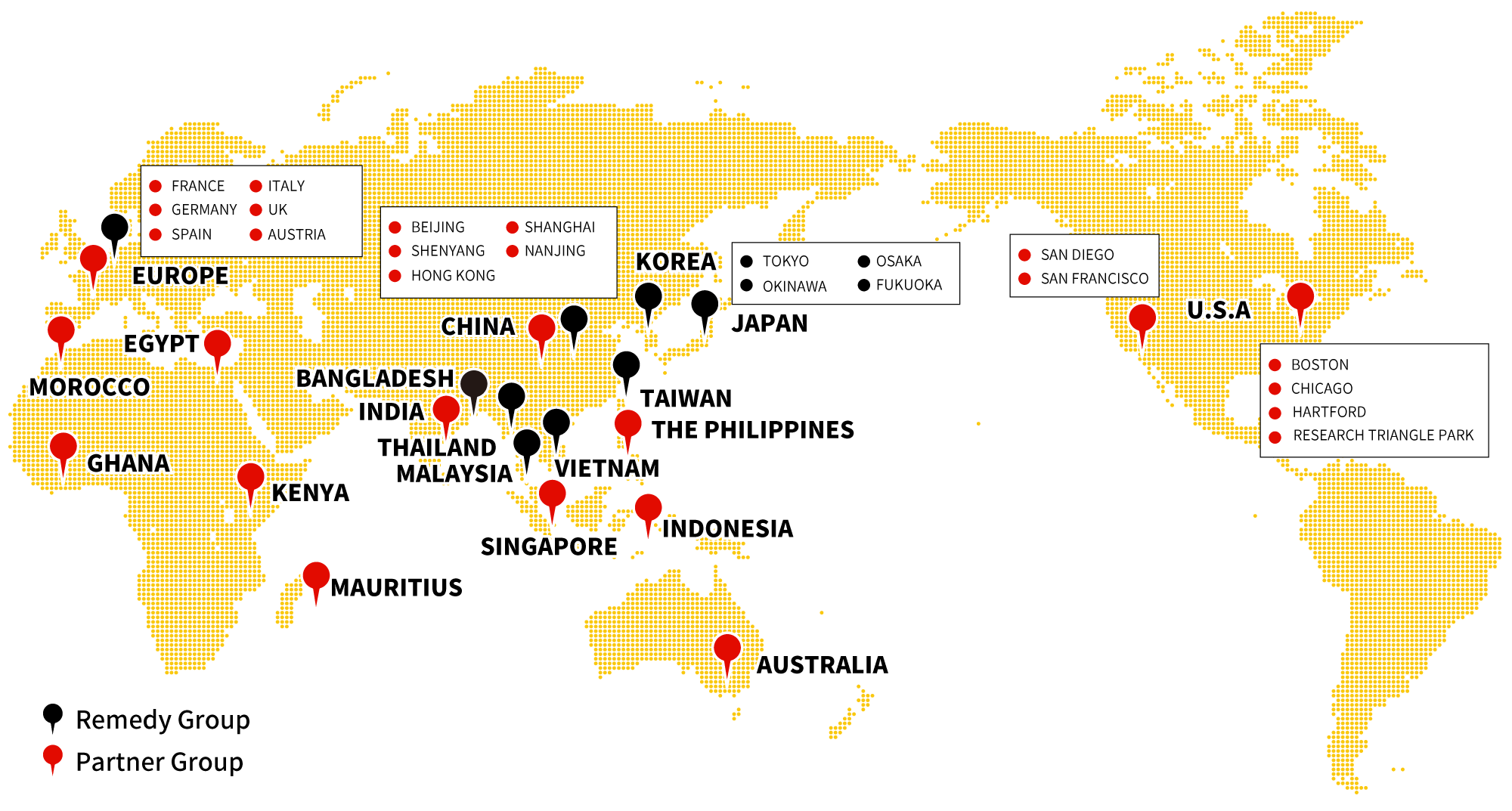
intellim provides full-scope support for regulatory affairs and various other drug development activities in Asian countries. We also propose development strategy on demand using our Global Network and experienced
resources.
See our service coverage in Asia.
For more information
If you have any comments, questions, please contact us.